U.S. officials gave full approval last week to Leqembi, a drug that moderately slows Alzheimer’s disease. The disease affects the brain causing memory loss and dementia.
The Food and Drug Administration (FDA) approval means the government’s health coverage, Medicare, and other health insurance plans will help pay for the drug’s cost.
The FDA approved Leqembi for patients with mild dementia and other effects caused during the early stages of Alzheimer’s.
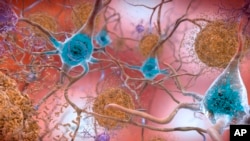
Leqembi is the first medicine shown to moderately slow the mental loss caused by Alzheimer’s disease. The drug helps remove proteins that stick together and damage the brain.
Final approval came after the FDA considered a study of 1,800 patients. The study showed the drug slowed losses in memory and decreases in thinking by about five months in those who received the medicine compared to those who received an ineffective substance.
The drug carries a warning for serious side effects, such as possible bleeding in the brain. The warning notes that these side effects are found in other drugs that target harmful brain proteins.
Last year, Medicare announced it would not pay for the drug unless the FDA gave it full approval. As a result, Alzheimer’s patients and people supporting them pushed for final approval.
A year’s supply of Leqembi costs about $26,500. It is injected every two weeks. Some experts worry the drug’s cost is too high. Medicare pays health care costs for about 60 million Americans aged 65 or older.
Medicare administrator Chiquita Brooks-LaSure confirmed last week that the program will begin paying for the drug now that it has full FDA approval.
But the government is also requiring patients who receive the drug to enter a program that will collect information about the drug’s safety and effectiveness.
Medicare “will cover this medication broadly while continuing to gather data that will help us understand how the drug works,” Brooks-LaSure said.
Some Medicare patients may have to pay for 20 percent of the cost of Leqembi. However, the amount will depend on the details of their insurance coverage plan.
Hospitals and medical clinics have said it might take time to get people started on the drug. Doctors will need to test patients for the sticky brain proteins targeted by Leqembi. Nurses need to learn how to give the drug, which needs to be administered by intravenous injection.
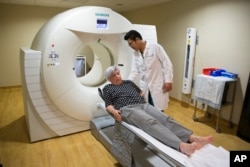
Patients also must undergo regular brain scans to look for bleeding or swelling. These brain images carry extra costs for hospitals.
Japanese drug-maker Eisai developed Leqembi. The drug is co-marketed with Biogen, a company based in the eastern U.S. state of Massachusetts.
Some Alzheimer’s experts say patients, or their families might not notice the moderate difference the drug makes. But federal health advisors said the difference was important enough for them to suggest in June that the FDA approve the drug.
I’m Andrew Smith.
Matthew Peronne wrote this story for The Associated Press. Andrew Smith adapted in for VOA Learning English.
_________________________________________________________________
Words in This Story
dementia –n. mental illness that causes a person to be unable to think clearly or understand the world
insurance –n. an agreement in which a person regularly pays a company and, in return, the company helps pay for costs that arise from some need like health care
intravenous –adj. something that is injected into the veins
scan –n. an image that shows something, like an organ, in three dimensions and is used by doctors to gather health information
swelling –n. when tissue becomes bigger than normal, gets deformed and holds too much fluid
________________________________________________________________
We want to hear from you.
We have a new comment system. Here is how it works:
- Write your comment in the box.
- Under the box, you can see four images for social media accounts. They are for Disqus, Facebook, Twitter and Google.
- Click on one image and a box appears. Enter the login for your social media account. Or you may create one on the Disqus system. It is the blue circle with “D” on it. It is free.
Each time you return to comment on the Learning English site, you can use your account and see your comments and replies to them. Our comment policy is here.





Forum